Question: 20.0 mL of NaCl (aq) reacts with 30.0 mL of Pb(C₂H₃O₂)₂ (aq). Knowing how to write the net ionic equation for a precipitation reaction is just the first step! Let's take a look at a concentration of ions question, where you calculate how much of each ion is present after a precipitation reaction. Make sure you include the phase of matter each compound is in. Omit the spectator ions and write the final net ionic equation of the given reaction. 
Write the complete ionic equation by dissociating soluble compounds into ions. It may already be balanced, but it also may not, so you always have to check. The best steps to follow when writing a net ionic equation are:įigure out which compounds are soluble and insoluble using solubility rules.īalance the chemical equation. We went over this in key topic 4.2, but let's do a quick overview! □ Usually, the question will tell you if the compound is soluble and which solution it’s soluble in. Table 4.1 is a table of solubility for common ions in water. You don’t need to know any other solubility rules for the AP, but it doesn’t hurt to be familiar with common soluble and insoluble compounds. Solubility RulesĪll sodium, potassium, and nitrate salts□ are soluble in water, so they aren’t precipitates. 
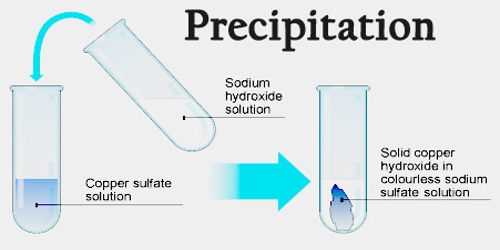
This solid product is called a precipitate. When ions in aqueous solutions react, they may produce an insoluble (undissolvable) or barely soluble solid ionic compound. We'll focus on precipitation reactions in this study guide! Precipitation reactions are chemical reactions in which two or more soluble reactants combine to form an insoluble product, which is known as a precipitate. These reactions involve the transfer of electrons from a reducing agent to an oxidizing agent and are characterized by a change in the oxidation state of the elements involved in the reaction.Ĭombustion reactions are a type of redox reaction and you've already learned about them! The specifics and must-know information about redox reactions will be covered later in this unit. Oxidation-reduction reactions, also known as redox reactions, are chemical reactions in which the atoms of one or more elements are oxidized (lose electrons) and reduced (gain electrons). For more about acid-base reactions, check out the next study guide. They often involve the transfer of a proton from a strong acid to a strong base, resulting in the formation of a salt and water. For now, here is a quick rundown of these three types of reactions:Īcid-base reactions are chemical reactions that involve the transfer of a proton from one molecule to another. Oxidation-reduction reactions are going to be reviewed in-depth in the rest of this unit. Throughout this unit's study guides, you've gotten a taste of net ionic equations and precipitation reactions, as well as titrations and acid-base reactions. One of the learning objectives of unit four in the AP Chemistry course is to "identify a reaction as acid-base, oxidation-reduction, or precipitation." Types of Reactions This unit is all about learning and identifying different chemical reactions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
